Jigsaw Puzzle : Ethanol and methoxymethane molecules
![]()

Jigsaw Puzzles from Science Photo Library
Ethanol and methoxymethane molecules
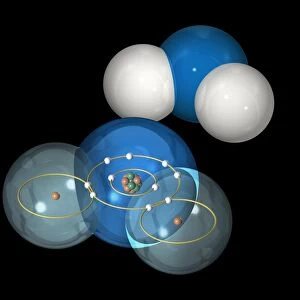
Ethanol and methoxymethane molecules. Molecular models of ethanol (CH3.CH2.OH, left) and methoxymethane (CH3.O.CH3). Both compounds contain the same atoms but in different arrangements. Carbon is black, hydrogen is white and oxygen is red. Ethanol (ethyl alcohol) is produced by the fermentation of sugar by yeast in alcoholic drinks. It is manufactured for use as a fuel and as a starting chemical for many other compounds. Methoxymethane (dimethyl ether) is used as an aerosol propellant and a fuel
Science Photo Library features Science and Medical images including photos and illustrations
Media ID 6294571
© ANDREW LAMBERT PHOTOGRAPHY/SCIENCE PHOTO LIBRARY
Atom Atoms B Alls Ball And Stick Bond Bonds Compound Compounds Ethanol Ethyl Alcohol Models Molecules Organic Sticks Structural Molecular
Jigsaw Puzzle (500 Pieces)
Discover the fascinating world of organic chemistry with our Media Storehouse Jigsaw Puzzles. This set includes two captivating puzzles featuring Ethanol and Methoxymethane molecules. Explore the intricate structures of these essential compounds, each made up of the same atoms but arranged differently. Engage your mind, challenge your problem-solving skills, and deepen your understanding of organic chemistry. A perfect addition to your home or classroom, these high-quality puzzles are sure to provide hours of educational fun for all ages.
500 piece puzzles are custom made in Australia and hand-finished on 100% recycled 1.6mm thick laminated puzzle boards. There is a level of repetition in jigsaw shapes with each matching piece away from its pair. The completed puzzle measures 40x51cm and is delivered packaged in an attractive presentation box specially designed to fit most mail slots with a unique magnetic lid
Jigsaw Puzzles are an ideal gift for any occasion
Estimated Product Size is 50.7cm x 40.3cm (20" x 15.9")
These are individually made so all sizes are approximate
Artwork printed orientated as per the preview above, with landscape (horizontal) or portrait (vertical) orientation to match the source image.
EDITORS COMMENTS
This print showcases the molecular models of ethanol and methoxymethane, two organic compounds with identical atoms arranged differently. The carbon atoms are represented by black spheres, hydrogen by white, and oxygen by red. Ethanol (CH3. CH2. OH), commonly known as ethyl alcohol, is produced through yeast fermentation of sugar in alcoholic beverages. However, its applications extend beyond drinks; it serves as a fuel source and a fundamental building block for numerous other chemical compounds. On the other hand, methoxymethane (CH3. O. CH3), also referred to as dimethyl ether, finds utility as an aerosol propellant and fuel. This compound shares the same atoms as ethanol but features an alternate structural arrangement. The intricate bond formations depicted in this image offer valuable insights into the fascinating world of organic chemistry. Molecular structures are visualized using ball-and-stick models that vividly illustrate how atoms connect to form compounds. Science Photo Library presents this remarkable print that not only highlights the beauty of these molecules but also provides a glimpse into their significance within various industries. It's worth noting that while both ethanol and methoxymethane have commercial applications such as fuel usage or aerosol propellants, this caption focuses solely on their scientific properties rather than any specific commercial use cases associated with them.
MADE IN AUSTRALIA
Safe Shipping with 30 Day Money Back Guarantee
FREE PERSONALISATION*
We are proud to offer a range of customisation features including Personalised Captions, Color Filters and Picture Zoom Tools
SECURE PAYMENTS
We happily accept a wide range of payment options so you can pay for the things you need in the way that is most convenient for you
* Options may vary by product and licensing agreement. Zoomed Pictures can be adjusted in the Cart.